Artificial intelligence has moved from experimental proofs-of-concept to operational deployment across the pharmaceutical manufacturing value chain. As of 2026, the global AI in pharmaceutical market is valued close to USD 4.5 billion, with manufacturing-specific applications capturing significant portion of this revenue.
For small and mid-size pharmaceutical companies, the transition to AI is no longer optional. These organizations face the same regulatory compliance obligations as large enterprises but operate with smaller budgets and leaner IT teams. Success in this landscape requires a shift from chasing algorithms to building foundational “Manufacturing Intelligence.”
The ROI Challenge: Why Only 22% of Leaders Scale Successfully
The potential for efficiency is high, yet only a small fraction of organizations realize AI ROI at scale. A 2026 Deloitte survey found that while 80% of organizations use generative AI in some capacity, only 22% of life sciences leaders have successfully scaled their AI initiatives. Just 9% reported significant financial returns.
The primary barrier to scaling is the “Digital Transformation Paradox.” Companies have invested millions in digital systems like ERP and MES, yet close to 70% of their data remains “dark”—physically present but analytically inaccessible. Poor data integration costs manufacturers 15-20% of their potential efficiency. For a mid-size manufacturer with USD 2 billion in revenue, this represents an annual loss of up to USD 400 million.
The Regulatory Landscape: Converging on Governance
Regulatory bodies have moved from observation to active governance. AI systems in GMP environments now face formal requirements for validation, traceability, and human oversight.
FDA: Risk-Based Credibility: The FDA’s “Computer Software Assurance” (CSA) guidance, finalized in September 2025, replaces exhaustive scripted testing with a risk-based approach. Additionally, the January 2025 draft guidance on AI support for regulatory decision-making establishes a credibility assessment framework that governs how AI data is used in drug applications.
EU GMP: Formalized Annex 22: In Europe, the introduction of Draft EU GMP Annex 22 represents the first dedicated framework for AI in manufacturing. It mandates strict adherence to ALCOA++ principles and requires human oversight for critical GxP activities like batch release.
Global Harmonization: The January 2026 joint publication of AI Practice Guiding Principles by the FDA, EMA, and Health Canada signals a future where a single validation package might satisfy multiple jurisdictions. Regulators emphasize “explainability,” discouraging black-box models in favor of architectures that allow quality teams to understand the rationale behind a decision.
Internal Adoption and Agentic Oversight by FDA: The FDA has moved beyond regulating AI to adopting it internally. In early 2026, the agency fully deployed Project Elsa, an internal generative AI platform with agentic capabilities. The FDA uses these agents to autonomously process historical data populations, identify behavioral patterns (such as closing large numbers of CAPAs before an inspection), and flag discrepancies between disparate systems like LIMS and MES logs.
This shift toward “real-time regulatory monitoring” means the FDA can identify process drifts and data integrity failures long before a physical site visit occurs. In April 2026, the FDA issued its first Warning Letter specifically citing a manufacturer for the improper use of AI agents in quality decisions without adequate human oversight.
High-Value AI Use Cases in Pharma Manufacturing
Specialized AI platforms are currently solving the technical bottlenecks that traditional systems cannot.
1. Genealogy: The Backbone of Traceability and Quality Assurance
Product genealogy (or material genealogy) is the detailed history and lineage of a product throughout its manufacturing journey. It tracks everything from raw materials through intermediates to the finished product and distribution. For Quality Assurance (QA) leaders, genealogy provides the comprehensive audit trail necessary to verify that quality standards have been met.
Challenges of Traditional Genealogy
Building a complete genealogy has traditionally been a slow, error-prone process. Data fragmentation across MES, LIMS, and paper logs creates silos that make manual reconciliation difficult. Legacy relational databases struggle to track the hierarchical, branching nature of material flows. For smaller firms, these complexities often stall genealogy initiatives.
How AI Automates Genealogy Construction
Modern AI-powered platforms, such as Mareana, simplify Genealogy implementation through several key functions:
- Automated Digitization: Pharma-specific OCR engines extract data from handwritten and printed records, reducing paper digitization efforts by up to 90%.
- Knowledge Graph Architecture: Unlike relational databases, AI-driven knowledge graphs automatically map relationships between materials, equipment, and outcomes. Years of batch history can be structured in hours rather than months.
- CDMO Integration: AI can ingest data from external manufacturing partners without requiring manual standardization, creating a unified view of the full lifecycle.
2. De-Risking Technology Transfer: Closing the “Know-How” Gap
Technology transfer is the complex migration of institutional knowledge, process expertise, and tacit know-how required to scale manufacturing. The traditional “push” model—where R&D hands over a static document package to manufacturing—frequently fails because it lacks the undocumented expertise (tacit knowledge) of the development team. This gap forces the receiving site to reverse-engineer the process, leading to delays, deviations, and millions in lost revenue.
Closing the Gap with Digital Twins
Manufacturing intelligence platforms like Mareana transform fragmented data into a complete, auditable digital record.
- Dynamic Digital Twins: By utilizing the Batch Genealogy module, AI creates an end-to-end visual map of all production data. This provides the new manufacturing site with an instant “digital twin” of the process, ensuring a deep understanding of historical behavior and process parameters.
- Ensuring Process Robustness (QbD): Successful tech transfer requires a deep understanding of the “design space.” Mareana’s Smart CPV (Continuous Process Verification) module uses predictive analytics to monitor raw materials and process parameters in real time. This ensures the process remains in a “state of control” at the new site, transitioning quality control from a reactive process to a proactive one.
Transforming Sponsor-CDMO Collaboration
The industry’s reliance on Contract Development and Manufacturing Organizations (CDMOs) makes tech transfer a mission-critical partnership. Traditional manual review methods create friction when discrepancies are found late in the release cycle.
- Unified Data Environments: A digital platform allows both the sponsor and CDMO to work from a shared source of truth with the help of a unified data platform.
- Exception-Only Workflows: Mareana’s Batch Release Copilot automates the reconciliation of batch records and trends. By flagging only deviations that require human attention, it accelerates batch release cycles and builds trust through transparency. This “first-time-right” approach is essential for accelerating drug pipelines and building resilient supply chains.
3. Review by Exception (RbE): Scaling Quality Oversight
Batch record reviews are a massive operational bottleneck. They are notoriously time-consuming and prone to human error due to reviewer fatigue and the sheer volume of data involved. While systems for capturing data have evolved, batch review decisions often still rely on a manual model designed for a paper-centric world.
Traditional Batch Review Software typically digitizes the paper format without automating the review logic. These systems operate in silos, which forces quality teams to manually reconcile data across fragmented platforms.
Review by Exception is a quality assurance strategy where reviewers only examine data that deviates from predefined standards. Instead of line-by-line audits of clean data, the system flags anomalies, allowing QA teams to focus their expertise on high-risk events.
Why Manual Review is Riskier Than You Think
Trusting a purely manual process involves several hidden risks that are difficult to defend under modern regulatory scrutiny:
- The Limit of Human Attention: Expecting a reviewer to maintain 100% accuracy across a 500-page batch record with hundreds of parameters is statistically unlikely.
- The “Invisible” Missing Data: Human eyes are naturally better at seeing incorrect values than finding what isn’t there, such as a missing signature or a blank field.
- Mathematical Human Error: Simple calculations on the shop floor are frequently done incorrectly and can easily be overlooked during a manual audit.
- Unstructured Data Blindness: Margin notes, strikeouts, and informal corrections contain critical context. During a long shift, these nuances are easily missed by human reviewers.
How AI Makes Review by Exception a Reality
The Mareana platform transforms paper-based data into a digital intelligence hub using three core pillars:
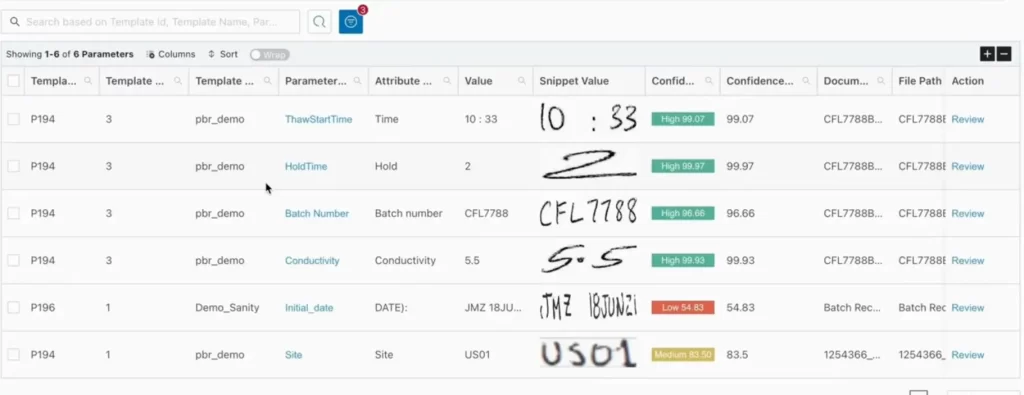
- AI-OCR for Actionable Data: Mareana digitizes everything from handwritten signatures to complex tables. The system uses “confidence scoring” to flag low-confidence snippets, ensuring that only data requiring a “human-in-the-loop” verification is surfaced.
- Automated Rule Verification: The platform performs real-time math verifications across multiple pages and checks values against the Master Batch Record (MBR). Correct calculations and within-range values are marked green, allowing the reviewer to skip redundant checks.
- AI Agents for Unstructured Exceptions: Specialized AI Agents detect “messy” data like margin notes. If a note says “Hold time extended by 15 minutes,” the system doesn’t just read it; it cross-references the SOP to verify if that extension is within the validated threshold. It then flags the note as a “Reviewed Exception” for final QA confirmation.
By implementing RbE, pharma manufacturers move from “looking for needles in haystacks” to an exception-only workflow that reduces total review time by up to 70%.
4. Smart Continuous Process Verification (CPV)
AI provides contextualized time-series data for real-time monitoring. By setting multivariate alerts, manufacturers can detect out-of-trend drifts three batches earlier than traditional manual monitoring. This facilitates the move toward real-time release.
5. Demand Forecasting and Shortage Mitigation
Drug shortages are a critical board-level priority. In early 2024, the U.S. reported over 323 active drug shortages. AI demand models that integrate external signals, such as hospital admission rates, can reduce forecast errors by 25–30%. In oncology cases, these tools have eliminated 80% of critical stockouts.
Strategic Recommendations for Pharma Manufacturers
Small and mid-size pharma companies must determine how to adopt AI in a compliant and ROI-positive way with limited resources.
- Prioritize Data Connectivity over Collection: Focus on building a “Manufacturing Intelligence” layer that breaks down silos between MES, LIMS, and ERP before deploying complex models.
- Adopt CSA Thinking: Update internal Standard Operating Procedures (SOPs) to reflect the FDA’s risk-based Computer Software Assurance framework.
- Manage the Model Lifecycle: Establish formal change control processes for retraining models and monitoring “model drift”—the performance degradation that occurs as process conditions change.
- Evaluate CDMO Capabilities: For asset-light organizations, the ability of a CDMO to share AI-ready data is a critical factor in vendor selection. Seek CDMO-agnostic platforms that can ingest paper or PDFs to achieve visibility without changing shop-floor workflows.
The technology for AI in pharma manufacturing is mature. Success over the next 24 months depends on the quality of an organization’s data foundations and the development of “trilingual” talent proficient in data science, life sciences, and GMP requirements.



 Learn more
Learn more



