Mareana CMC is a state-of-the-art digital SaaS product specifically designed to meet the unique needs of pharmaceutical and biotech firms of all sizes during the crucial pre-commercial stage of drug development. By integrating every phase of the CMC lifecycle, Mareana CMC ensures compliance, enhances operational efficiency, and significantly speeds up market entry, allowing firms to move swiftly from development to delivery.
Business Benefits
| Task Description | % Improvement |
|---|---|
| Cross-functional Collaboration | 50% |
| Task Description | % Improvement |
|---|---|
| Scale-up Studies | 56% |
| Cross-functional Collaboration | 88% |
| Troubleshooting and Problem-solving | 50% |
| Task Description | % Improvement |
|---|---|
| Batch Record Review | 70% |
| Internal Audits | 50% |
| Deviation and CAPA Management | 63% |
| Complaint Handling | 50% |
| Data Integrity Oversight | 50% |
| Task Description | % Improvement |
|---|---|
| Protocol Development | 17% |
| Study Design and Planning | 17% |
| Data Management | 33% |
| Safety Monitoring | 17% |
| Statistical Analysis | 33% |
| Report Writing | 25% |
| Documentation and Record Keeping | 25% |
| Task Description | >% Improvement |
|---|---|
| Document Management | 33% |
Key Features
Mareana CMC offers a suite of advanced features designed to optimize every stage of the drug development process. Each feature is purpose-built to enhance functionality, reduce manual effort, and ensure regulatory compliance, providing a comprehensive solution for pharma and biotech firms so that they can Make Confidently, Release Confidently, and Comply Confidently.
Mareana™ CMC
Enterprise Digital CMC Solution
Automate data management & compliance reporting across the entire drug development cycle.
GxP-Compliant & Purpose-Built
Meet GxP requirements with our purpose- built and validated solution. With built-in data integrity measures, ensure the accuracy and reliability of data for analytics and reporting.
End-to-End Data Traceability
Get end-to-end traceability of data from raw material to finished product attributes.
User-Friendly Interface
Minimize training time and maximize productivity with the best-in-class design enabling ease of use.
Enhanced Scalability
Scale effortlessly within a business with your growing data and process needs. Our cloud-based application supports both small-scale projects and large, multi-site operations
Automated Data Processing
Extract and contextualize data from paper batch records and other third- party data sources using Al & automation framework.
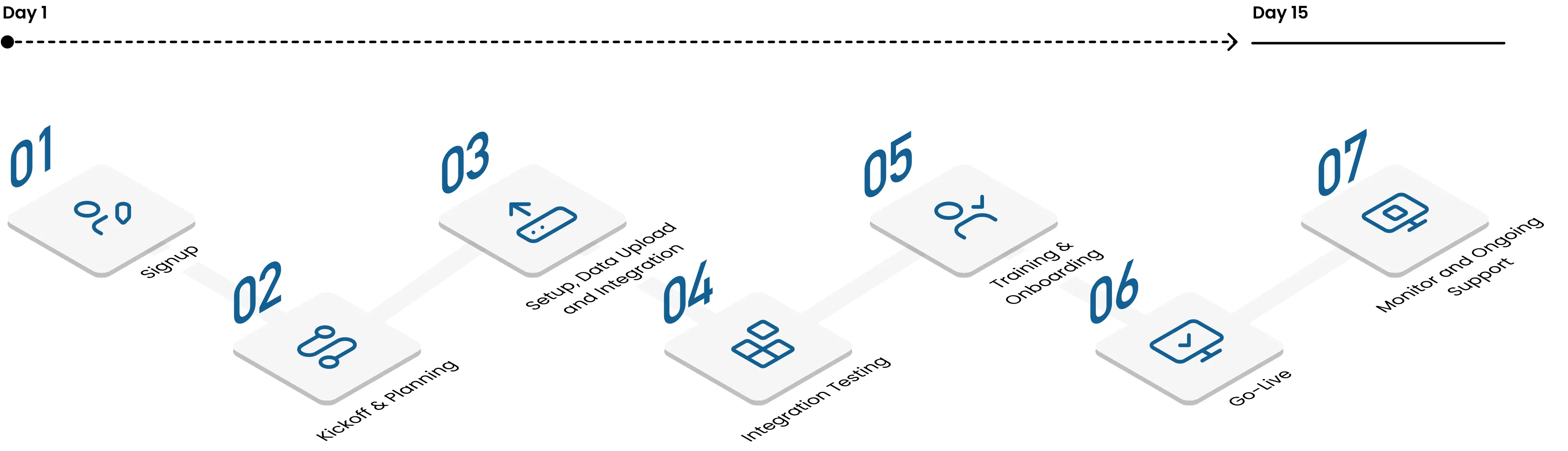
Typical Deployment Timeline
Customers Who Trust Us






FAQs About Mareana CMC
Mareana CMC is a state-of-the-art digital SaaS product designed to streamline Chemistry, Manufacturing, and Controls (CMC) manufacturing processes. Mareana CMC ensures compliance with regulatory standards, automatically integrates data from different source systems and data types (including paper batch records), provides data analytics with an integrated data science studio, and simplifies reporting, ultimately enhancing efficiency and reducing time to market.
Our platform is built in accordance with global pharmaceutical standards, including FDA and EMA guidelines. It is fully validated and GMP compliant.
Currently the only data source we support are Paper batch records and soon we will have measures for seamless integration with a wide range of existing systems and platforms, ensuring minimal disruption to your operations.
Data security is a top priority for us. Mareana CMC employs advanced security measures, including encryption and regular security audits, to ensure your data is protected at all times.
We offer comprehensive support including training sessions, a dedicated customer service team, and regular updates. Our goal is to ensure you maximize the benefits of Mareana CMC in your operations.
Mareana CMC stands out due to its comprehensive compliance management, Paper batch record data handling, integrated data analytics capability, user-friendly interface, and dedicated customer support. Our solution is specifically tailored to the needs of the pharmaceutical manufacturing industry, making it uniquely suited to address your challenges.
We provide extensive training for new users, including online tutorials, videos, and personalized training sessions, to ensure your team is fully equipped to use the product effectively.
Stay Ahead with
Mareana™
Take your pharmaceutical manufacturing to the digital age with an AI-driven platform that empowers you to Make Confidently, Release Confidently, and Comply Confidently.

Contact Us
Would you like to learn more about our pharmaceutical manufacturing solutions? Fill out the form below and one of our experts will reach out to you promptly.


